
The Bohr radius constant is based on Bohr's model of the atom. When it loses a certain amount of energy, it falls to a smaller, lower shell. When an electron absorbs enough energy, it moves to a larger, or higher, shell. These distances are represented by spheres, called shells, surrounding the nucleus. The Bohr atom included negatively charged electrons orbiting the nucleus at specific median distances. In the Rutherford atom of atomic theory, electrons orbit the nucleus in circular paths.Īnother physicist, Niels Bohr, revised Rutherford's atomic model in 1913. He was the first to suggest that atoms are like miniature solar systems, except that instead of gravity acting as the attractive force, opposing electrical charges serve that function. The physicist Ernest Rutherford developed an early model of the atom in 1912. The only neutral atom with no neutrons is the hydrogen atom. Carbon 14 is a less common, radioactive carbon isotope.Carbon 12 is the most common, non-radioactive isotope of carbon.For example, the following are two isotopes of the carbon atom: An isotope for an element is specified by the sum of the number of protons and neutrons. 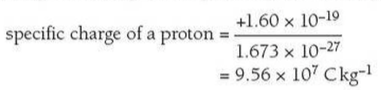
An ion with extra electrons has a negative charge and is called an anion and an ion deficient in electrons has a positive charge and is called a cation.Ītoms having the same number of protons but different numbers of neutrons represent the same element and are known as isotopes of that element. Normally, atoms have equal numbers of protons and electrons, giving them a neutral charge.Īn ion is an atom with a different number of electrons than protons and is electrically charged. Protons have a positive charge and electrons a negative charge. Protons and electrons have equal and opposite charges. Because the number of neutrons in an atom can vary, there can be several different atomic weights for most elements. For most purposes, the atomic weight can be thought of as the number of protons plus the number of neutrons.  
Protons and neutrons make up the nucleus of an atom and the electrons orbit.Įlectrons contribute only a tiny part to the mass of the atomic structure, however, they play an important role in the chemical reactions that create molecules. The atomic mass or weight is measured in atomic mass units. The total mass of an atom, including the protons, neutrons and electrons, is the atomic mass or atomic weight. The mass of a proton or neutron increases when the particle attains extreme speed, for example in a cyclotron or linear accelerator.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
